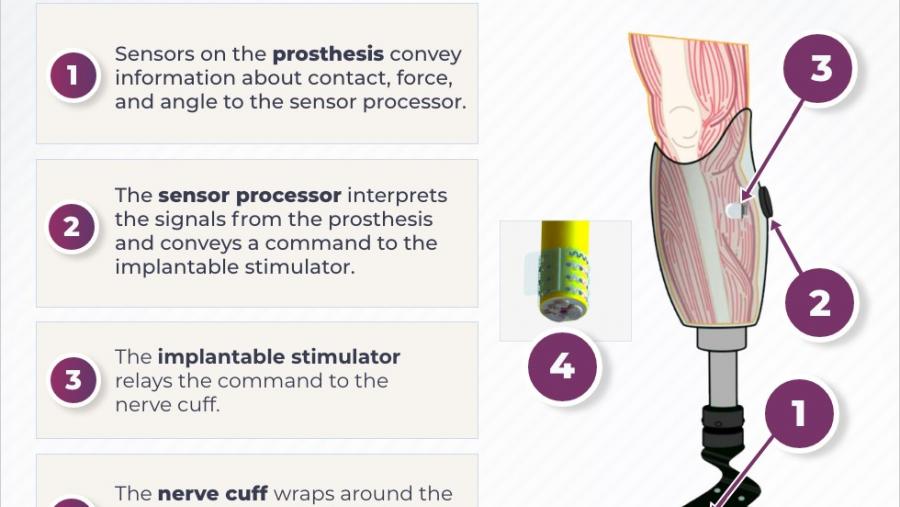
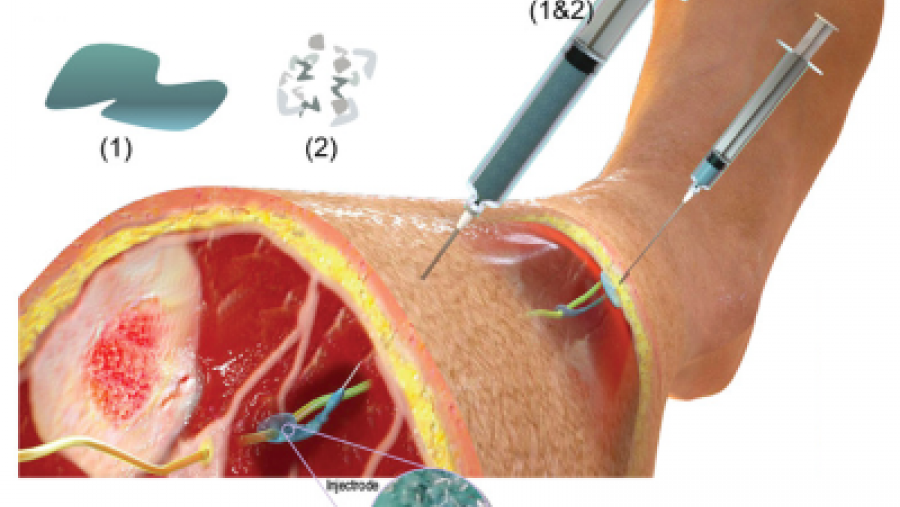
Translating Discoveries into Effective Devices to Treat Pain
Overview
The Research Need
Opioid misuse is a major factor in the development of opioid use disorder. Advances in technology show promise for identifying new, safer pain management options to improve quality of life and reduce reliance on opioids. Innovative technology designs may also reduce side effects compared to medication-based strategies. Several effective devices for pain have already received approval or clearance from the U.S. Food and Drug Administration.
About the Program
This program fosters the development of next-generation medical devices to diagnose and treat both pain and opioid use disorder. The research supports preclinical development and translational research efforts to advance promising technologies to readiness for testing in clinical trials.
The program also promotes development of groundbreaking, safe, and effective medical devices via the NIH Blueprint MedTech incubator program. This program supports and de-risks the development of medical devices targeting nervous system disorders, including chronic pain and opioid use disorder, from early development stages through first-in-human clinical trials.
Another focus of the program is to support interdisciplinary team science and academic-industry partnerships to understand how medical devices work to relieve pain. This new body of knowledge will uncover new pain biomarkers and optimize clinical approaches to enhance pain relief for patients.
Research Examples
Research examples supported by this program include:
- Developing an injectable electrode to be inserted into the spinal cord to treat chronic back pain
- Using image-guided technologies such as magnetic resonance imaging (MRI) or electroencephalograms to aim focused ultrasound at specific brain regions to interrupt pain circuits related to chronic pain conditions, such as sickle cell disease pain
- Developing a non-invasive electroacupuncture device to treat pain associated with irritable bowel syndrome
- Developing nanoparticles for intravenous delivery of ketamine using focused ultrasound in specific regions of the brain to treat chronic osteoarthritic pain while avoiding off-target effects
- Using MRI-guided, focused ultrasound to treat head and neck cancer pain
- Using multisite, closed-loop deep brain stimulation to treat chronic neuropathic pain
- Conducting a mechanistic clinical trial to uncover mechanisms underlying pain relief by transcutaneous auricular neurostimulation
- Developing and validating pain-associated outcome measures in non-rodent mammalian models
- Baylor College of Medicine – Texas
- Carnegie-Mellon University – Pennsylvania
- Duke University – North Carolina
- Massachusetts General Hospital – Massachusetts
- Micro-Leads, Inc. – Massachusetts
- Novaflux, Inc. – New Jersey
- Ripple, LLC – Utah
- Stanford University – California
- University of California, Los Angeles – California
- University of California, San Francisco – California
- University of Michigan – Michigan
- University of Pittsburgh – Pennsylvania
- University of Texas Medical Branch – Texas
- University of Utah - Utah
- University of Virginia – Virginia
- University of Wisconsin–Madison – Wisconsin
- Vanderbilt University Medical Center – Tennessee
Participating NIH Institutes, Centers, and Offices
- National Cancer Institute (NCI)
- National Center for Advancing Translational Sciences (NCATS)
- National Center for Complementary and Integrative Health (NCCIH)
- National Eye Institute (NEI)
- National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS)
- National Institute of Biomedical Imaging and Bioengineering (NIBIB)
- National Institute of Dental and Craniofacial Research (NIDCR)
- National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK)
- National Institute of Neurological Disorders and Stroke (NINDS)
- National Institute on Aging (NIA)
- National Institute on Alcohol Abuse and Alcoholism (NIAAA)
- National Institute on Drug Abuse (NIDA)
- NIH Common Fund
View Other Research Programs in This Focus Area
- Development and Optimization of Non-Addictive Therapies to Treat Pain
- Discovery and Validation of Novel Targets for Safe and Effective Treatment of Pain
- Preclinical Screening Platform for Pain (PSPP)
- Restoring Joint Health and Function to Reduce Pain Consortium (Re-JOIN)
- Translational Research to Advance Testing of Novel Drugs and Human Cell-based Screening Platforms to Treat Pain and Opioid Use Disorder
- Integrated Basic and Clinical Team-Based Research in Pain
 U.S. Department of Health & Human Services
U.S. Department of Health & Human Services